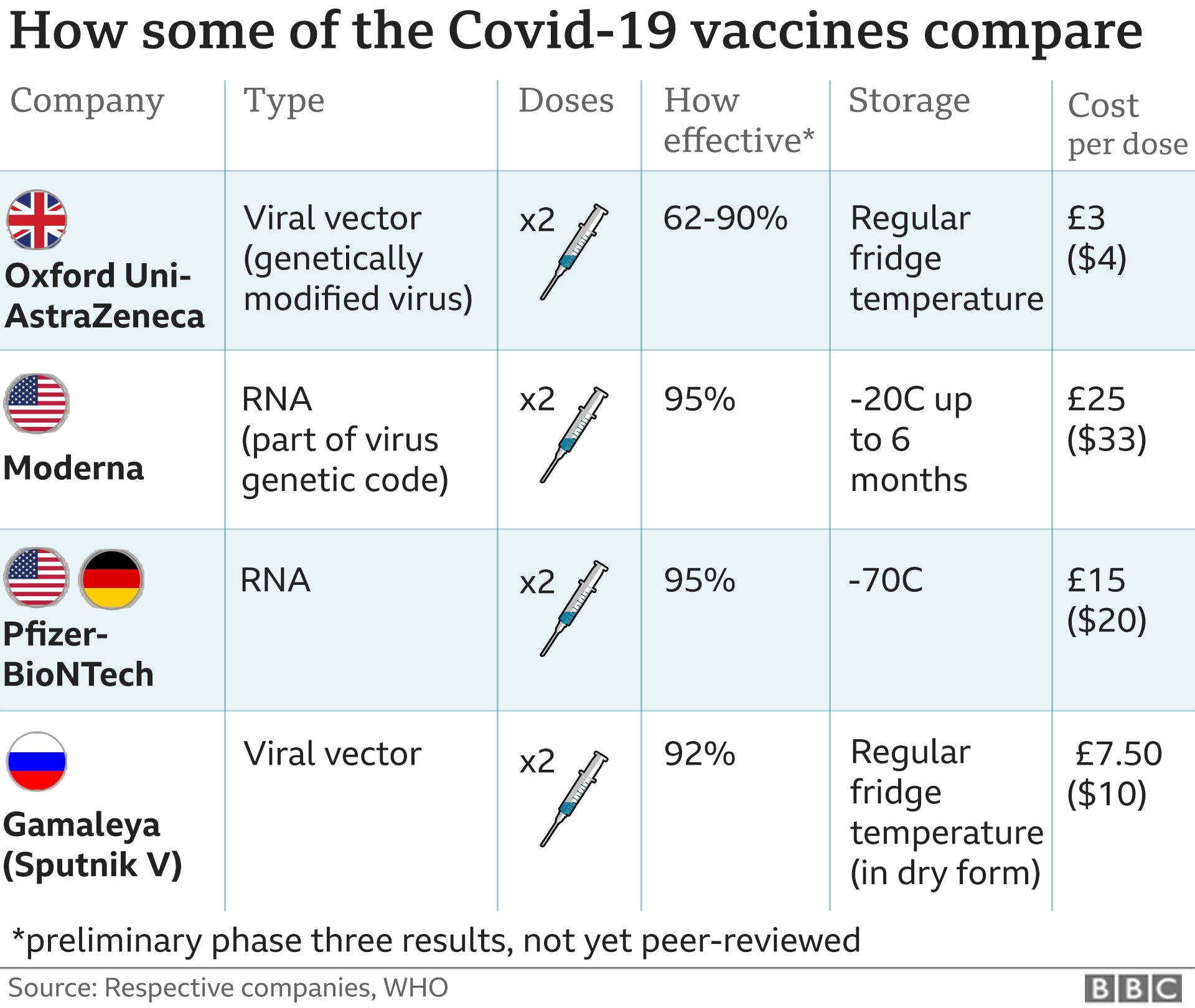
The World Health Organization (WHO) recommends an interval of three to four weeks between doses. The initial course consists of two doses. It must be thawed to room temperature and diluted with normal saline before administration. The vaccine is supplied in a multidose vial as "a white to off-white, sterile, preservative-free, frozen suspension for intramuscular injection". The vaccine is used to reduce morbidity and mortality from COVID-19.  
The Pfizer–BioNTech COVID-19 vaccine is used to provide protection against COVID-19, caused by infection with the SARS-CoV-2 virus, by eliciting an immune response to the S antigen. In September 2022, the European Union authorized both the BA.1 and the BA.4/BA.5 (tozinameran/famtozinameran) booster versions of the bivalent vaccine for people aged twelve years of age and older. In September 2022, the BA.1 version of the bivalent vaccine (Comirnaty Original/Omicron BA.1 or tozinameran/riltozinameran) was authorized as a booster for use in the United Kingdom for people aged twelve years of age and older. In August 2022, a bivalent version of the vaccine (Pfizer-BioNTech COVID-19 Vaccine, Bivalent) was authorized for use as a booster dose in individuals twelve years of age and older in the United States. Distribution and storage is a logistical challenge because the vaccine needs to be stored at extremely low temperatures. As of 30 March 2021, Pfizer and BioNTech aimed to manufacture about 2.5 billion doses in 2021. Īs of September 2021, more than 1.5 billion COVID-19 vaccine doses have been shipped by the company worldwide. On 23 August 2021, the Pfizer–BioNTech vaccine became the first COVID-19 vaccine to be approved in the United States by the Food and Drug Administration (FDA) for those aged sixteen years and older. It is authorized for use at some level in the majority of countries. In December 2020, the United Kingdom was the first country to authorize its use on an emergency basis. The vaccine is the first COVID‑19 vaccine to be authorized by a stringent regulatory authority for emergency use and the first cleared for regular use. Reports of serious side effects, such as allergic reactions, are very rare and no long-term complications have been reported. The most common include mild to moderate pain at the injection site, fatigue, and headaches. Most side effects are mild to moderate in severity and are gone within a few days. An interim analysis of study data showed a potential efficacy of 91.3% in preventing symptomatic infection within seven days of a second dose and no serious safety concerns. Ĭlinical trials began in April 2020 by November 2020, the vaccine entered phase III clinical trials, with over 40,000 people participating. Initial advice indicated that vaccination required two doses given 21 days apart, but the interval was later extended to up to 42 days in the US, and up to four months in Canada. It is composed of nucleoside-modified mRNA (modRNA) encoding a mutated form of the full-length spike protein of SARS-CoV-2, which is encapsulated in lipid nanoparticles. The vaccine is given by intramuscular injection. It is authorized for use in people aged five years and older in some jurisdictions, twelve years and older in some jurisdictions, and for people sixteen years and older in other jurisdictions, to provide protection against COVID-19, caused by infection with the SARS-CoV-2 virus. For its development, BioNTech collaborated with American company Pfizer to carry out clinical trials, logistics, and manufacturing. The Pfizer–BioNTech COVID-19 vaccine ( INN: tozinameran), sold under the brand name Comirnaty, is an mRNA-based COVID-19 vaccine developed by the German biotechnology company BioNTech.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
